
CasNo: 149-32-6
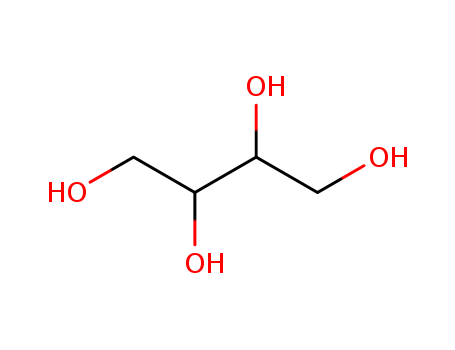
MF: C4H10O4
Appearance: white crystals or powder
|
Characteristics |
The sweetness of erythritol is low, the sweetness of erythritol is only 60%-70% of sucrose, the entrance has a cool taste, the taste is pure, and there is no post-bitterness. It can be used in combination with high-intensity sweeteners to inhibit its Undesirable flavors of high-intensity sweeteners. Erythritol has high stability, is very stable to acid and heat, and has high acid and alkali resistance. It will not decompose and change at temperatures below 200 °C, and will not undergo Maillard reaction to cause discoloration. The heat of dissolution of erythritol is high: erythritol has an endothermic effect when dissolved in water, and the heat of dissolution is only 97.4kJ/kg, which is higher than the endothermic degree of glucose and sorbitol, and has a cooling feeling when eating. The solubility of erythritol at 25 °C is 37% (W/W). With the increase of temperature, the solubility of erythritol increases, and it is easy to crystallize and separate out crystals. Erythritol is very easy to crystallize, but it will not absorb moisture in a 90% humidity environment. It is easy to be crushed to obtain a powdery product, which can be used on the surface of food to prevent food from absorbing moisture and deteriorating. |
|
Production Methods |
Erythritol is a starch-derived product. The starch is enzymatically hydrolyzed into glucose which is turned into erythritol via a fermentation process, using osmophilic yeasts or fungi (e.g. Moniliella pollinis, or Trichosporonoides megachiliensis). |
|
Biotechnological Production |
The synthesis of erythritol is rather difficult. One of the possibilities is the catalytic reduction of tartaric acid with Raney nickel, which does, however, also produce threitol, a diastereomere of erythritol that requires separation of both. Threitol may be isomerized which increases the yields of erythritol. Another chemical synthesis starts from butane-2-diol-1.4 which is reacted with chlorine in aqueous alkali to yield erythritol-2-chlorohydrin and can be hydrolyzed with sodium carbonate solution. Synthesis from dialdehyde starch in the presence of a nickel catalyst at high temperatures is also possible. Owing to the special physiological properties of erythritol, commercial interest increased with the discovery of an increasing number of microorganisms able to produce this substance. Today, the commercial production of erythritol is apparently only based on fermentation. Erythrytitol fermentations mostly use osmophilic yeasts. Based on regulatory submissions for commercial production, T. megachiliensis, M. pollinis, and Y. lipolytica are used. It is also claimed that P. tsukubaensis and Aureobasidium sp. are used for commercial production. Erythritol-producing microorganisms often produce other polyols such as ribitol. Nevertheless, some strains had a rather high yield of erythritol. A two-step fermentation of C. magnoliae on 400 g/L glucose resulted in a 41 % conversion rate and a productivity of 2.8 g/Lh. M. pollinis cultivated on glucose and several nitrogen sources yielded erythritol concentrations up to 175 g/L with a conversion rate of 43 %. Oxygen limitation resulted in ethanol formation, and nitrogen limitation in strong foaming. A mutant gave even better yields. Aerobically on glucose cultured P. tsukubaensis KN 75 produced 245 g/L of erythritol with an especially high yield of 61 %. The productivity was 2.86 g/Lh. Scale-up from 7-L laboratory fermenter to 50,000-L industrial scale resulted in productivities similar to the laboratory value. |
|
Flammability and Explosibility |
Notclassified |
|
Pharmaceutical Applications |
Erythritol is a naturally occurring noncariogenic excipient used in a variety of pharmaceutical preparations, including in solid dosage forms as a tablet filler, and in coatings. It has also been investigated for use in dry powder inhalers.It is also used in sugar-free lozenges,and medicated chewing gum.Erythritol can also be used as a diluent in wet granulation in combination with moisture-sensitive drugs. In buccal applications, such as medicated chewing gums, it is used because of its high negative heat of solution which provides a strong cooling effect. Erythritol is also used as a noncaloric sweetener in syrups; it is used to provide sensorial profile-modifying properties with intense sweeteners; and it is also used to mask unwanted aftertastes. Erythritol is also used as a noncariogenic sweetener in toothpastes and mouthwash solutions. |
|
Biochem/physiol Actions |
Allelic variation of the Tas1r3 gene affects behavioral taste responses to this sugar alcohol, suggesting that it is a T1R3 receptor ligand. |
|
Safety |
Erythritol is used in oral pharmaceutical formulations, confectionery, and food products. It is generally regarded as a nontoxic, nonallergenic, and nonirritant material. However, there has been a case report of urticaria caused by erythritol. The low molecular weight of erythritol allows more than 90% of the ingested molecules to be rapidly absorbed from the small intestine; it is not metabolized and is excreted unchanged in the urine. Erythritol has a low caloric value (0.8 kJ/g). The WHO has set an acceptable daily intake of ‘not specified’ for erythritol. Erythritol is noncariogenic; preliminary studies suggest that it may inhibit the formation of dental plaque. In general, erythritol is well-tolerated; furthermore, excessive consumption does not cause laxative effects. There is no significant increase in the blood glucose level after oral intake, and glycemic response is very low, making erythritol suitable for diabetics. LD50 (mouse, IP): 8–9 g/kg LD50 (rat, IV): 6.6 g/kg LD50 (rat, oral): >13 g/kg |
|
storage |
Erythritol has very good thermal and chemical stability. It is nonhygroscopic, and at 25°C does not significantly absorb additional water up to a relative humidity (RH) of more than 80%. Erythritol resists decomposition both in acidic and alkaline media and remains stable for prolonged periods at pH 2–10.(10) When stored for up to 4 years in ambient conditions (20°C, 50% RH) erythritol has been shown to be stable. |
|
Purification Methods |
meso-Erythritol crystallises from distilled water or absolute EtOH and is dried at 60o in a vacuum oven. It sublimes at 110o in a high vacuum. It is optically inactive. [Jeans & Hudson J Org Chem 20 1565 1955, IR: Kuhn Anal Chem 22 276 1950, Beilstein 1 IV 2807.] |
|
Incompatibilities |
Erythritol is incompatible with strong oxidizing agents and strong bases. |
|
|
|
|
Sources |
Naturally occurring in plants, fermented foods, and biological fluids of humans and animals. |
|
Chemical Composition and Structure |
Erythritol has a chemical formula C4H10O4 and is a four-carbon sugar alcohol. It occurs as a white anhydrous crystalline powder. |
|
Mechanism of Action |
Erythritol is absorbed quickly and excreted unchanged in urine. It does not affect glucose or insulin levels and may promote gut hormone release.[1] |
|
History and Development |
Traces of erythritol found in blackstrap molasses residue in the 1950s. Commercial production began in the early 2000s. FDA and EU approvals granted in the early and mid-2000s. In 2019, the global erythritol market transactions reached 70,400 tons equivalent to US$198.3 million, projected to rise to US$310 million by 2026.[1] |
|
Definition |
ChEBI: The meso-diastereomer of butane-1,2,3,4-tetrol. |
|
General Description |
meso-Erythritol, belonging to the class of sugar alcohols, is identified in a variety of food products, fruits, vegetables, beverages and dietary supplements. It is known as a low glycemic food additive and plays an important role as a sweetener for diabetic patients, since it does not have glycemic or insulinemic effect due to its ability to not get metabolized but get absorbed in the small intestine. It is also reportedly used as a sugar substitute in toothpaste, chewing gums, confectionery food products, etc. |
InChI:InChI=1/C4H10O4/c5-1-3(7)4(8)2-6/h3-8H,1-2H2
Alcohols have a wide range of applicabil...
Sugar alcohols are of great importance f...
The invention relates to a process for p...
Abstract: In the present investigation, ...
cis-2,3-epoxy-1,4-butanediol

1,2,3,4-butanetetrol
| Conditions | Yield |
|---|---|
|
With water; at 100 ℃; for 9h;
|
99% |
1,4-butenediol

1,2,3,4-butanetetrol
| Conditions | Yield |
|---|---|
|
With potassium permanganate; dihydrogen peroxide; In water; acetone; at 0 ℃; for 2h; Temperature; Inert atmosphere; Green chemistry;
|
60% |
D-threose
glucose
D-Xylose
<2R-(2α(2R*(2'S*,3a'R*,4'R*,7'R*,7a'R*),3R*),3aα,4β,7β,7aα)>-2,4-Bis<(octahydro-7,8,8-trimethyl-4,7-methanobenzofuran-2-yl)oxy>-1,3-butandiol
(+/-)-erythronic acid
3,4-dihydroxy-2-phenylhydrazono-butyraldehyde phenylhydrazone
1,3:2,4-di-O-benzylidenerythritol
tetrakis-O-(toluene-sulfonyl-(4))-erythritol